What is Galvanized Steel? - Carbon steel is inherently susceptible to corrosion, which means it must be protected by some other metallic coating that is less corrosive. Carbon steel sheet is called “galvanized” when coated with a thin layer of pure zinc. This coating provides two types of corrosion protection for the steel: “barrier protection” (sealing the surface from air and water) and “sacrificial protection” or “galvanic” protection—especially at scratches and cut edges.
This is an electrolytic process whereby the base metal is protected, but at the expense of the coating. The zinc “sacrifices” itself to protect the underlying layer of steel.
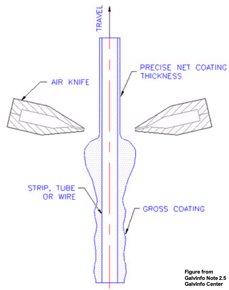 |
Air Knife Operation - controls the thickness of the coating during the molten metal bath |
How is the Steel Coated?
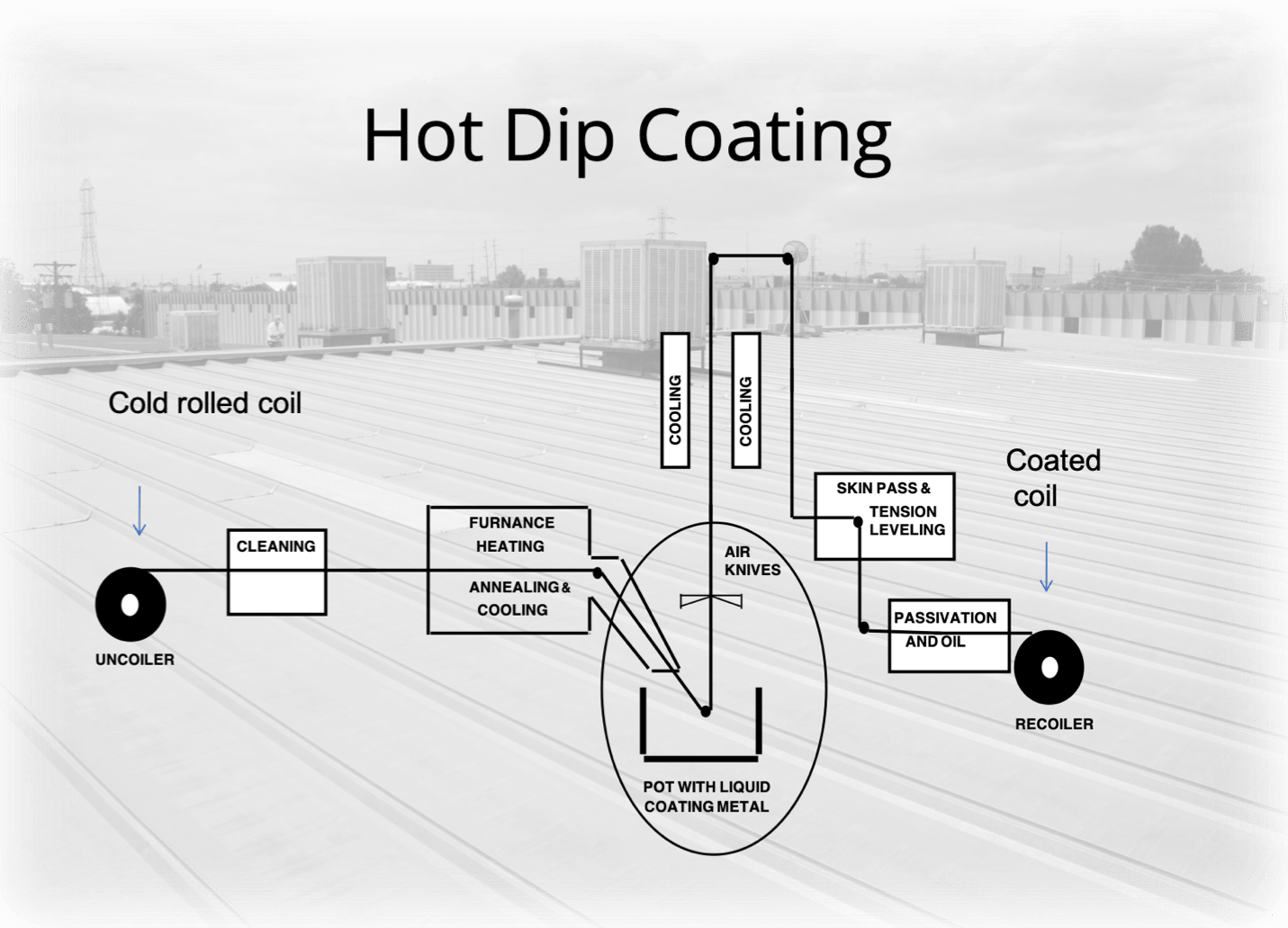
Protective coatings are most often applied to the steel coil at the producing mill. This “continuous hot-dip galvanizing” process takes place after the steel has been thoroughly cleaned, degreased, placed into an acid bath (pickling), rinsed, air dried and pre-heated.
A Bath of Molten Metal
The steel coil is then given its protective bath of molten metal. This step must be performed at the appropriate temperature (typically 600°F(+)) in order to ensure a metallurgical bond between the coating and the steel.
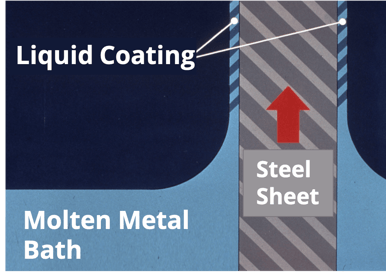
During the molten metal bath, “air knives” control the thickness of the coating. These “knives” are pneumatic squeegees (operation by air or gas under pressure) that evenly disperse the coating as the steel emerges from the bath. The cooling process is closely monitored so as not to traumatize the steel coil. The entire hot-dip process operates at an approximate speed of about 800 linear feet per minute – resulting in 4,800 square feet per minute of coil being coated.
Limits of Galvanized Steel
By design, the galvanized steel zinc coating goes away as it “sacrifices” itself to protect the underlying base metal. Different environments will degrade the coating at different speeds (e.g. high acidity, marine, etc.). This is why the life of the steel is directly related to the thickness of the coating AND the elements to which it is exposed.
Galvanized Metal Service Life
Zinc-coated steel is not usually warranted against corrosion because of the variables mentioned. The zinc gradually diminishes due to natural oxidation: Over time, the base steel becomes exposed, as the barrier wears away, causing corrosion of the base metal to begin. As mentioned above, wetter conditions exacerbate this wearing down of the coating. Drier climates and steep sloped roofs fare much better. In other words, the defining factor in service life reduction is not really the amount of rainfall, but rather the amount of time that rain lingers on the roof accelerating the galvanic process.
 |
From hot-dip coating to the breakthrough invention of Galvalume®, steel coatings have revolutionized the metal roofing industry. Learn more and download the complete white paper. |
______________________________________________________________________________
New Technologies Take Hold
The process of using pure zinc as a protective coating has become less popular over the last several decades. The invention of better coating technologies have shifted the industry. 
The most popular carbon steel coating is now aluminum/zinc alloy (55% AlZn). Known as Galvalume®, by most US domestic producers, it combines the superior barrier protection of aluminum with the sacrificial properties of zinc. Galvalume offers superb weathering protection while still providing galvanic protection from corrosion. Scratch and cut-edge protection of Galvalume is also very good – this is the aluminum and zinc combining to prevent exposed-edge corrosion and progression.
What is Galvalume?
The tradename “Galvalume®” combines the words galvanized (zinc) and aluminum. This coating for steel was co-invented in the late 1960s by Angelo Borzillo and Jim Horton (of Bethlehem Steel). The two researchers developed a coating that combined the corrosion-resistance of aluminized steel with the benefits of galvanized steel applied by the same continuous hot-dip method, but now at temperatures above 1200ºF. And so, Galvalume was born. The specific make-up consists of 55% Aluminum, 43.4% zinc by weight, and 1.6% silicon. (By volume the coating is about 80% aluminum.) The addition of the silicon serves to assist with coating adhesion to the steel during application, roll-forming and other fabrication activities.
BIEC International Inc. has licensed its 55% AlZn coating technology to multiple producers so it can be found under additional tradenames (e.g. Aluzinc®, Zincalume®, Zintro-alum™, and others). Its exterior uses extend to residential, commercial, industrial and agricultural walls, roofs and soffits. Additional uses include appliances, unexposed automotive parts, mufflers, ducts, garden tools, culverts, framing, and more.
Galvalume Steel Service Life
The expected service life (as documented by industry) of Galvalume for roofing applications is many times that of galvanized steel—in the 60-100+ year range, and most producers warrant the material for 25 years depending upon the thickness applied and the environment. (Salt-spray environments are excluded.)
 What Are the “Do’s and Don’ts” of Using Galvalume?
What Are the “Do’s and Don’ts” of Using Galvalume?
Strong alkalis or acids will attack this coating, as will copper salts and graphite. Do not mark with graphite pencil or allow contact with wet concrete or mortar. Severe corrosive effects may result
Contact with or run-off from copper surfaces is also to be avoided. Treated lumber contains copper salts and should never be used on 55% Al-Zn surfaces. Lumber of any kind is porous and traps moisture against the coating so that it cannot air-dry. This leads to premature corrosion. Like galvanized, 55% Al-Zn likes to breath and any kind of capillary entrapment can lead to unwanted corrosive effects.

Other areas of concern when using Galvalume include some agricultural settings. Direct contact or heavy fumes from animal waste, fertilizers, and animal confinement areas can be aggressive towards the finish of Galvalume due to the high ammonia content. Galvalume may retain cosmetic stains, such as footprints and handprints that occur during handling and installation. Short-term protection against these marks can be in the form of a thin acrylic coating (but only for unpainted applications). The acrylic is applied at the mill and eventually wears away but protects Galvalume against staining during handling and installation.
 |
 |
| Lumber of any kind is porous and traps moisture against the coating so that it cannot air-dry. | The image above illustrates how premature corrosion can occur due to using wood as a mounting structure. Moisture becomes trapped - causing corrosion. |
Word De-Construction
Word of the Week: Galvanized
PRONUNCIATION: \ ˈgal-və-ˌnīz -ed
PART OF SPEECH: adjective
SENTENCE: // The zinc coating on galvanized steel gradually diminishes due to natural oxidation.
Where Does the Word Galvanized Come From?: In the 1770s, an Italian physician and physicist named, Luigi Galvani, began exploring electrical currents and their impact on nerve impulses. His research was unfounded as he attempted to prove that animal tissue had innate electrical impulses.
Nevertheless, it is from Galvani that we acquired the word “galvanized”. A current of electricity produced through chemical action is galvanismo in Italian. Through the years the word morphed into galvanism in English and later into galvanize during the 19th century. Ultimately, it has transformed into its most popular use as galvanized – meaning to cover metal with zinc or zinc-rich alloy as a rust protection utilizing an electrolytic process. The word “galvanize” is also used outside of metallurgy as a verb to describe a catalyst from bringing together two or more things, especially people in relationship or association with unique synergisms.
Word De-Construction
Word of the Week: Galvalume®
PRONUNCIATION: \ ˈgal-və-lo͞om
PART OF SPEECH: noun
SENTENCE: // Galvalume® is a hot-dip coated steel that marries the barrier corrosion-resistance of aluminized steel with the sacrificial protection of galvanized steel.
Where Does the Word Galvalume Come From?: The company (Bethlehem Steel) that invented a new alloy coating for carbon steel thin sheet used mostly in cladding applications and consisting primarily of aluminum and zinc in a specific formulation chose this word as their trademark, combining the words “galvanize” and “aluminum”. The alloy was patented and licensed to more than 50 other producers worldwide. The license granted to these producers also included the right to use the “Galvalume” trademark. Some producers elected to use the mark, and others decided to create their own mark. The alloy, therefore, is also know by other trademarks such as “Zincalume” and others.
Why Use S-5! Clamps?
All S-5! clamps and related hardware are manufactured with non-corrosive 300 series stainless steel. Over 1,000 hours of ASTM B-117 salt fog testing helps ensure that our attachments cause no corrosion on painted Galvalume or galvanized panels. Learn more about our extensive testing here.

S-5! Technical Support: Should I be Concerned with Metallurgical Compatibility?
Blog Title: What is Swarf?
S-5! Manufacturing – Why is it the Most Trusted Name in the Metal Construction Industry?


